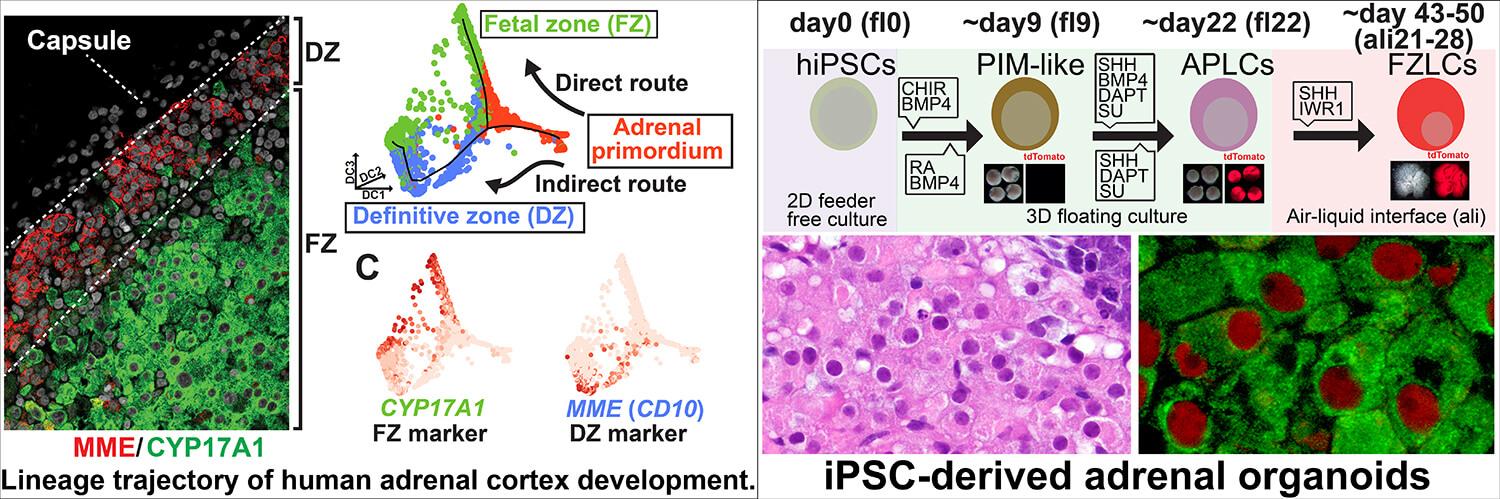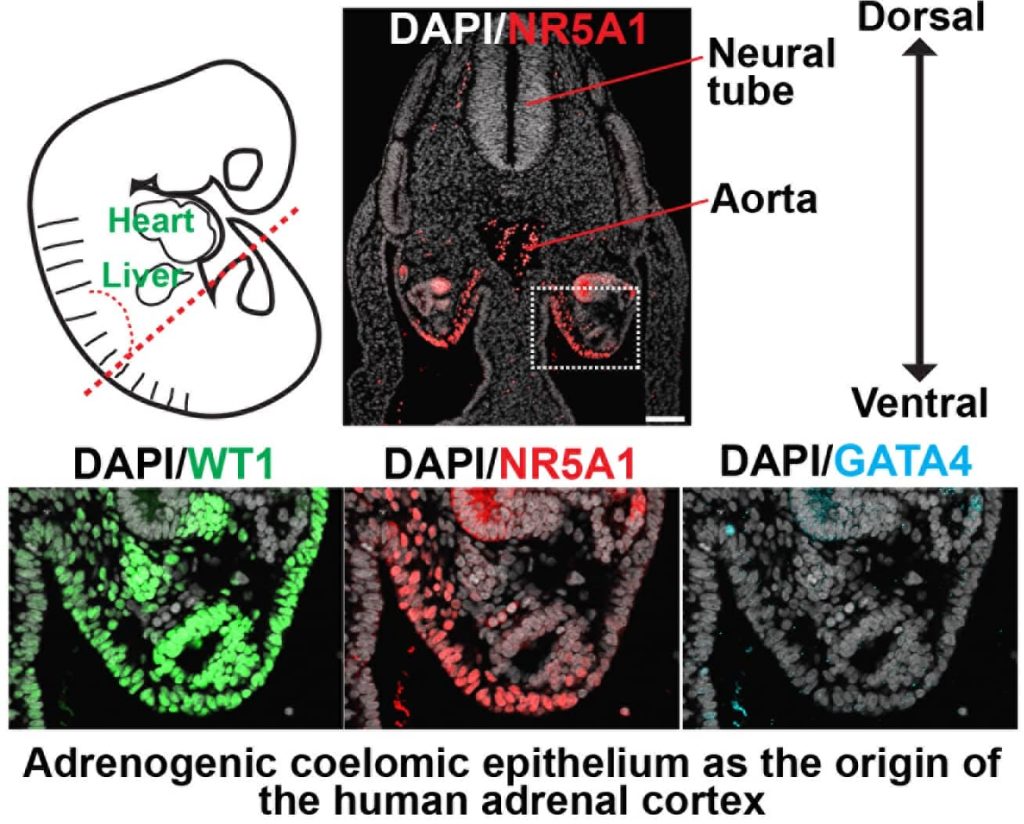
Reconstitution of human adrenal gland and its therapeutic applications
The adrenal cortex is the major endocrine hub for steroid hormone production, and thus regulates a wide array of critical physiologic functions essential to human life, including immune and stress responses, sexual maturation and electrolyte balance. However, the molecular mechanisms driving human adrenal cortex development and steroid biosynthesis are poorly understood due to the paucity of appropriate model systems. We recently developed the first human induced pluripotent stem cells (iPSCs)-derived fetal adrenal organoid system that recapitulates normal functional development and steroidogenesis. Using this system, we will identify cellular and signaling mechanisms and gene regulatory networks orchestrating human adrenocortical development and steroid biosynthesis to provide key insight into the pathophysiology of primary adrenal insufficiency and adrenal androgen excess and to serve as a stepping stone for eventual regenerative therapy for patients with a wide range of adrenal diseases.